Gold Testing with Electrical Conductivity Measurement Test
18 August 2023

In an increasingly volatile global financial system, fraught with counterparty risks and economic uncertainties, holding physical gold has long been extolled as a prudent strategy. Gold, often called the "crisis commodity," is a hedge against inflation, currency devaluation, and other financial calamities. It is a tangible asset that you can hold in your hand, free from the risks associated with digital currencies, stocks, or bonds, which are subject to the whims of market fluctuations and the stability of financial institutions.
However, owning physical gold is not without its challenges. One of the most significant risks that gold owners face is the authenticity of their precious metals. Counterfeit gold bars and coins are not uncommon, and the consequences of holding inauthentic gold can be financially devastating. Therefore, verifying the authenticity of your gold is not just an option; it's an imperative.
This brings us to the critical question: How can one ascertain the authenticity of physical gold? In past articles, we have introduced ultrasound testing and X-ray fluorescence testing as frequently used non-destructive testing methods. This article explores how Electrical Conductivity Measurement Tests (ECM) work and why it is increasingly used for gold testing.
Electrical Conductivity Measurement test principles and how it works
Electrical conductivity is a measure of a material's ability to conduct an electric current. In simpler terms, it quantifies how easily electrons can flow through a substance when subjected to an electric field.
Good conductors of electricity, like metals, have high electrical conductivity, allowing for easy flow of electrons, while insulators like rubber and wood have low conductivity, hindering the movement of electrons.
The basic principle behind electrical conductivity lies in the atomic or molecular structure of the material. In metals, for instance, the outer electrons of the atoms are loosely bound and are free to move throughout the metal lattice. These "free electrons" are what enable the flow of electric current.
When an electric field is applied across a conductor, it exerts a force on these free electrons, causing them to move and thereby creating an electric current. The greater the density of free electrons and the less resistance they encounter, the higher the material's electrical conductivity.
Conductivity is commonly expressed in units of Siemens per meter (S/m), millisiemens per meter (mS/m), or Megasiemens per meter (MS/m) and is the reciprocal of electrical resistivity.
Factors affecting conductivity in metals
Various factors can affect electrical conductivity, including temperature, impurities, and the presence of defects in the material. For example, as a metal's temperature increases, its atomic lattice's vibrations also increase, causing more frequent collisions between the free electrons and the lattice. This results in increased resistance and decreased conductivity.
Similarly, impurities and defects can disrupt the smooth flow of electrons, affecting the material's overall conductivity. Metals are often alloyed with other elements to improve certain properties like strength or corrosion resistance. However, these alloying elements can disrupt the uniform lattice structure of the metal, impeding the flow of free electrons and thus reducing conductivity.
For instance, while pure gold is highly efficient at conducting electricity, its electrical conductivity can be notably reduced by introducing even minor amounts of other metals like copper or silver. In a similar vein, the electrical conductivity of copper can also be diminished when adulterated with impurities.
How conductive are precious metals like gold, silver and platinum?
Precious metals like gold, silver, and platinum are known for their high electrical conductivity, making them valuable for their aesthetic appeal and various industrial applications.
Among these, silver stands out as the most electrically conductive element, offering a conductivity of approximately 61.35 MS/m. Its exceptional ability to conduct electricity makes it ideal for use in electrical contacts, conductive inks, and high-frequency RF connectors.
Gold, while not as conductive as silver, is still an excellent conductor of electricity, with a conductivity of around 44.7 MS/m. One of the advantages of gold over silver is its resistance to tarnish and corrosion, which makes it highly desirable in applications where reliability over long periods is crucial, such as in aerospace and medical devices.
Platinum, on the other hand, has lower electrical conductivity compared to both silver and gold, with values around 9.4 MS/m. However, platinum’s high melting point and excellent resistance to corrosion make it useful in specialized applications like high-temperature sensors and electrodes.
How an Electrical Conductivity Measurement Test is conducted
At Silver Bullion, we use the Sigma Analytics Precious Metal Verifier to test gold, silver, and platinum accurately. The device is known for its ease of use, and quick results, making it a popular choice among jewelers, pawn shops, and precious metal dealers.
ECM tests are also useful because they are a non-destructive testing method, unlike a fire assay or an acid test. The latter requires a few drops of acid to be put on the gold item. Therefore, an ECM test for real gold will not result in damaging precious bullion gold coins, bars, and gold jewelry.
The Precious Metal Verifier is also portable, allowing you to test gold at home.
The following steps are generally followed when conducting an Electrical Conductivity Measurement Test:
Preparing the test sample
It is important to clean the precious metal sample with a clean, lint-free cloth to remove any surface impurities, oils, or coatings that could interfere with the test results.
Any plastic coin capsule holding the precious metal bar or coin should be removed because the ECM Test will not yield an accurate result.
Calibrating the Precious Metal Verifier
Before starting the ECM Test, it's important to calibrate the Sigma Analytics Precious Metal Verifier. The testing device usually comes with calibration samples, and following the manufacturer's guidelines ensures that the device will provide accurate readings.
Testing with the Precious Metal Verifier
Once turned on, different testing profiles can be selected for testing the sample. For example, there are inbuilt testing profiles for precious metals of different fineness:
Gold: Pure gold .999+, 91.7% 22K bal Cu, 90% bal Cu, American Eagle, Krugerrand, 98.6%, Britannia 90-12
Silver: 99.99% Pure, 99.9% Pure, 92.5% Sterling, 90% US pre-1900, 90% US pre-1945, 90% US Coin 1960, 96% Britannia, 80% Canadian Maple Leaf
Other: Platinum, Palladium, Rhodium
With the testing profile selected, place your sample on the circular onboard sensor if the sample has a minimum diameter of 24 mm and thickness of 1 mm.
Use the small testing wand if your sample has a minimum diameter of 8 mm and thickness of 0.8 mm. For larger testing samples, use the large wand, which is capable of testing items with a minimum diameter of 18 mm and thickness of 0.8 mm.

Understanding the Precious Metal Verifier’s results
The Precious Metal Verifier sends a small electrical current through the metal and measures the resistance encountered. It then calculates the electrical conductivity based on this resistance and produces a result on its digital display to indicate if the sample is genuine gold, silver, or platinum.
- Sample is consistent with the selected testing profile
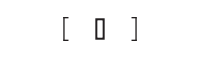
A result anywhere in the brackets means the sample is consistent with the selected test profile.
- Sample is slightly inconsistent with the selected testing profile
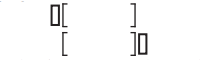
If the box cursor falls outside of the brackets, to either the right or to the left, that indicates that the sample is slightly inconsistent with the selected alloy type.
- Sample is inconsistent with the selected testing profile

The further the box cursor is outside of the brackets, the less consistent the sample’s characteristic resistivity is with the selected alloy type and the more questionable the sample is.
- Sample is highly inconsistent with the selected testing profile

An arrow indicates that a sample is off the scale in that direction. This indicates the sample is highly inconsistent with the selected alloy type.

Never rely on only one type of test
Relying solely on one method for gold testing can be risky and potentially inaccurate in determining if the gold is real, as each gold testing technique has its own set of limitations and vulnerabilities. For instance, while electrical conductivity tests are excellent for quick, non-destructive assessments, they may not be as effective in identifying fake gold-plated items that have a thick layer of gold.
X-ray fluorescence tests are mainly a surface testing method and may not detect fake gold bars that are thickly gold-plated.
The best way to test for real gold is to subject the gold piece to two or more different gold testing methods. While counterfeiters may easily create fake gold bars or fake gold jewelry to mimic one material property of gold, it becomes increasingly difficult or near impossible to mimic real gold for two or more material properties.
The tables below show that gold's density is very similar to that of tungsten, a base metal. Therefore, a fake gold bar that is made of tungsten will weigh almost identically on a weighing scale. However, gold's material properties for celerity, and electrical conductivity are very different from that of tungsten.
Celerity, Density & Electrical Conductivity of Precious Metals
| Metal | Celerity (m/s) | Density (g/cm³) | Electrical Conductivity (MS/m) |
| Gold | 3240 | 19.30 | 44.7 |
| Silver | 3650 | 10.49 | 61.35 |
| Platinum | 3830 | 21.06 | 9.4 |
Celerity, Density & Electrical Conductivity of Tungsten & Lead
| Metal | Celerity (m/s) | Density (g/cm³) | Electrical Conductivity (MS/m) |
| Tungsten | 5220 | 19.25 | 18.52 |
| Lead | 2160 | 11.34 | 5 |
A fake gold item made of tungsten can be detected easily if an additional test like an ultrasound test or electrical conductivity test is also performed. Using at least two methods to test gold is definitely the better way to determine if a sample is real gold.
The Safe House Also Performs Electrical Conductivity Measurement Tests
The Safe House has performed ECM tests for countless precious metals entering the vault’s premises. We conduct our non-destructive precious metals tests, including tests for density, material thickness using ultrasound, surface composition using X-ray fluorescence, and electrical conductivity, from our testing labs at The Reserve. Once completed, bullion tests can be accompanied by a precious metals testing report.
We have many years of experience testing gold and testing gold jewelry. Our gold testing machines are well able to detect different fake gold items, from heavy gold plated bars, and gold filled with other metals, to gold alloys.
Please do not hesitate to contact us should you have any questions.